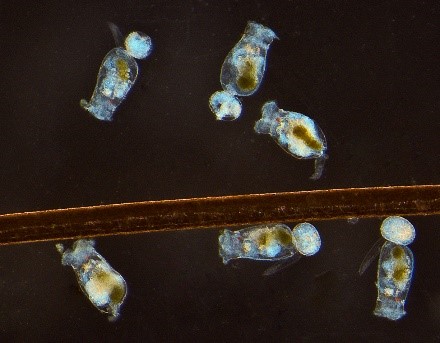
Sustainable Seafood
Feeding a hungry world; restoring depleted marine populations
The world’s population is slated to grow by 2.4 billion people by 2050. According to the Food and Agriculture Organization of the United Nations, we must produce 70 percent more food to meet impending world hunger needs. As some wild fish stocks are decreasing, aquaculture is the practical solution to produce food sustainably, as well as to help replenish depleted fisheries. Aquaculture also can provide significant value to local economies. Reducing our country’s reliance on imported seafood would reduce the U.S. trade deficit and provide “blue tech” jobs to revitalize our working waterfronts.
Our research team and its collaborators have played a leadership role for over 35 years in developing innovative replenishment solutions with no negative biological impacts. Breeding and raising fish from minuscule eggs to a market-ready size is a complicated process. It requires invention and significant knowledge from many fields of science, including the understanding of sophisticated hatchery infrastructure to ensure an optimal environment for fish to flourish.
Whether we’re producing white seabass and halibut for release into the wild, or yellowtail, mussels or seaweed for the seafood market we are preserving and renewing wild stocks by providing supplemental sources of plants and animals that are produced sustainably.
Help us feed the world, replenish fish populations and provide economic value to American coastal communities.

Mark A. Drawbridge, M.S.
Program Leader
Primary Research Areas
Fisheries Replenishment
Healthy, sustainable fisheries are essential for reliable seafood supply and thriving recreational angling opportunities. However, many threats exist in the marine environment that have led to declining fish abundances. Aquaculture offers a solution – cultured juveniles can be released to grow naturally in the wild and help restore fish populations.Hubbs-SeaWorld Research Institute is one of the leading proponents of this type of fisheries replenishment in the United Sates. Since the early 1980’s, we have been developing the aquaculture technology and expertise to release millions of white seabass (Atractoscion nobilis), a species highly prized by seafood consumers and anglers in California. Through the support of Dorothea Laub, Paul and Norma Fruchbom and Chevron Corporation, HSWRI established the Dick Laub Fisheries Replenishment Program in 2014. This program aims to build on the approach developed for white seabass (i.e. fisheries replenishment using releases of cultured fish) and apply it to other marine finfish species in southern California.Our scientists work across a wide variety of disciplines (e.g. nutrition, health, genetics) and collaborate with fishing communities, resource managers and academia, among others, to ensure that fisheries replenishment work is carried out to the highest environmental standards.
For more on white seabass replenishment: Ocean Resources Enhancement and Hatchery Program (OREHP)
For more on halibut replenishment: The Dick Laub Fisheries Replenishment Program
Farm to Table
Aquaculture products can be used to fill a wide range of marketing niches but they are most commonly associated with the production of food, similar to agriculture on land. Most of the species of fish cultured commercially in the United States were originally developed for stocking purposes. Once the main hurdles of spawning and larval rearing are overcome, the process of growing fish out for direct human consumption becomes a rather simple process from a biological and technological standpoint. Thus, demonstrating the feasibility of a direct farm-to-table approach was a logical transition for us to pursue from a research standpoint. Having a species that can be successfully stocked into the ocean and also raised directly for food is a winning combination.
Integrated Multi-Trophic Aquaculture
Our research team has long sought to diversify our portfolio of cultured organisms beyond finfish. Recently we received two grants that is allowing us to do just that. The first project is focused on a combination of fish and seaweed, while the second will add invertebrates like mussels to that formula. Here we describe the first project where the primary goal is to establish a land-based pilot program for rearing native marine macroalgae that compares stand-alone culture with an integrated multi-trophic (IMTA) approach. To execute this project we have teamed up with a company that specializes in seaweeds – Sunken Seaweed.
Primary Research Areas – In Depth
Fish Nutrition
Understanding the role of nutrition for each life stage of an organism under culture is an important factor for producing high quality fish for either replenishment or seafood production. We have conducted extensive nutrition research on adults, larvae, and juveniles to be able to influence not only survival but to improve larval and juvenile quality. At HSWRI we have begun studying broodstock nutrition and have the capabilities to run replicated trials on large pelagic species such as California yellowtail (Seriola dorsalis). Larval nutrition is perhaps the most challenging because this life stage is so small and fragile, and organ systems are still developing. Marine fish larvae are fed one or more live prey that are enriched to enhance their nutritional value before the larvae are weaned onto formulated diets. This dietary regime and transition of food types adds to the complexity. From a sustainability standpoint, the juvenile (growout) stage is very important because of the large biomass involved that requires substantial amounts of food. Historically, diets for carnivorous species consisted of large proportions of fish meal and oil to meet the dietary protein and lipid requirements of the species. With aquaculture growing so rapidly, those natural resources will become scarcer and more expensive. Thus, the quest for alternative sources of oil and protein is an area we have studied extensively.
Published Research
Lopez, L.M., Torres, A.L., Durazo, E., Drawbridge, M.A. and D.P. Bureau. 2006. Effects of lipid on growth and feed utilization of white seabass (Atractoscion nobilis) fingerlings. Aquaculture 253:557-563.
Lopez, L.M., E., Durazo, M.T. Viana, M.A. Drawbridge, and D.P. Bureau. 2009. Effect of dietary lipid levels on performance, body composition and fatty acid profile of juvenile white seabass, Atractoscion nobilis. Aquaculture 289: 101–105
Durazoi, E., A.C. Cruz, L.M. Lopez, J.P. Lazo, M. Drawbridge and M.T. Viana. 2010. Effects of digestible protein levels in isonitrogenous diets on growth performance and tissue compositions of juvenile Atractoscion nobilis. Aquaculture Nutrition. 16(1): 54-60(7)
Jirsa, D.O., D.A. Davis, and M.A. Drawbridge. 2010. Development of a practical soy-based diet for white seabass, Atractoscion nobilis. North American Journal of Aquaculture. 72:332–337.
Vizcaíno-Ochoa, V., J.P. Lazo, B. Barón-Sevilla, and M. Drawbridge. 2010. The effect of dietary docosahexaenoic acid (DHA) on growth, survival and pigmentation of California halibut Paralichthys californicus larvae (Ayres, 1810). Aquaculture. Vol. 302, no. 3-4, pp. 228-234.
Galaviz, M.A., García-Gasca, A., Drawbridge, M.A., Álvarez-González, C.A., and L.M. López, 2011. Ontogeny of the digestive tract and enzymatic activity in white seabass, Atractoscion nobilis, larvae. Aquaculture 318:162-168.
Jirsa, D., A. Davis, K. Stuart, and M. Drawbridge. 2011. Development of a practical soy-based diet for California yellowtail, Seriola lalandi. Aquaculture Nutrition 17:e869-e874.
Jirsa, D., Deng, D.F., Wang, W., Hung, S., Davis, D.A. and M.A. Drawbridge. 2012. The effects of dietary lipid levels on performance and heat shock protein response of juvenile white seabass, Atractoscion nobilis. Aquaculture Nutrition 19:227-232.
Trushenski, J., B. Mulligan, D. Jirsa, and M. Drawbridge. 2013. Sparing fish oil with soybean oil in feeds for white seabass: effects of inclusion rate and soybean oil composition. North American Journal of Aquaculture 75:305-315.
Jirsa, D., G.P. Salze, F.T. Barrows, D.A. Davis, and M. Drawbridge. 2013. First-limiting amino acids in soybean-based diets for white seabass Atractoscion nobilis. Aquaculture 414-415:167-172.
Jirsa, D., D.A. Davis, F.T. Barrows, L.A. Roy, and M. Drawbridge. 2014. Response of white seabass to practical diets with varying levels of protein. North American Journal of Aquaculture. 76(1):24-27.
Jirsa, D., D.A. Davis, G.P. Salze, M. Rhodes, and M. Drawbridge. 2014. Taurine requirement for juvenile white seabass (Atractoscion nobilis) fed soy-based diets. Aquaculture 422-423:36-41.
Jirsa, D O; Stuart, K R; Salze, G P; Rhodes, M A; Davis, D A; Drawbridge, M.A. 2014. Limiting Amino Acids in Practical Diets for California Yellowtail, Seriola lalandi World Aquaculture Society. Journal, Journal of the World Aquaculture Society; 45(6): pp. 681-690
Trushenski, J., A.N. Rombenso, M. Page, D. Jirsa, and M. Drawbridge. 2014. Traditional and fermented soybean meals as ingredients in feeds for white seabass and yellowtail jack. North American Journal of Aquaculture. 76(4): 312-322.
Jirsa, D., Barrows, F. T., Hardy, R. W., and M Drawbridge. 2015. Alternative protein blends as a replacement for fish meal in diets for white seabass, Atractoscion nobilis. Aquaculture nutrition, 21(6), 861-867.
Buentello, A., D. Jirsa, F.T. Barrows, and M.A. Drawbridge. 2015. Minimizing fishmeal use in juvenile California yellowtail, Seriola lalandi, diets using non-GM soybeans selectively bred for aquafeeds. Aquaculture 435: 403–411.
Hawkyard, M., Stuart, K., Langdon, C. and M.A. Drawbridge. 2015. The enrichment of rotifers (Brachionus plicatilis) and Artemia franciscana with taurine-liposomes and their subsequent effects on the larval development of California yellowtail (Seriola lalandi). Aquaculture Nutrition. DOI: 10.1111/anu.12317.
Rombenso, A.N., Trushenski, J.T., Jirsa, D. and M. Drawbridge. 2015. Successful Fish Oil Sparing in White Seabass Feeds Using Saturated Fatty Acid-Rich Soybean Oil and 22:6n-3 (DHA) Supplementation. Aquaculture. pp. 176-185 DOI: 10.1016/j.aquaculture.2015.05.041
Rombenso, A.N., Trushenski, J.T., Jirsa, D. and M. Drawbridge. 2016. Docosahexaenoic acid (DHA) and arachidonic acid (ARA) are essential to meet LC-PUFA requirements of juvenile California Yellowtail (Seriola dorsalis). Aquaculture 463:123–134
Rotman, F., K. Stuart, and M. Drawbridge. 2016. Effects of taurine supplementation in live feeds on larval rearing performance of California yellowtail Seriola lalandi and white seabass Atractoscion nobilis. Aquaculture Research, 1–8, doi:10.1111/are.12964
Lopez, L.M., J.O. Soto, I.T Escamilla, M.F. Ibarra, L. Ochoa, M. Drawbridge, and H. Peres. 2016. Evaluation of carbohydrate-to-lipid ratio in diets supplemented with Bacillus subtilis probiotic strain on growth performance, body composition and digestibility in juvenile white seabass (Atractoscion nobilis, Ayres 1860). Aquaculture Research, 47, 1864–1873
Salze, G. P., Davis, D. A., Jirsa, D. O. and Drawbridge, M. A. 2017. Methionine Requirement for Juvenile White Seabass, Atractoscion nobilis, Using Nonlinear Models. J World Aquaculture Soc. doi:10.1111/jwas.12413
Stuart, K., Hawkyard, M., Barrows, F., Rust, M., and Drawbridge, M. 2017. Evaluation of dietary taurine concentrations in microparticulate diets provided to larval California yellowtail (Seriola dorsalis) postlarvae. Aquaculture Nutrition (1) 1-11.
Salze, G. P., Stuart, K. R., Jirsa, D. O., Davis, D. A. and Drawbridge, M. A. 2018. Quantitative Dietary Taurine Requirement for California Yellowtail, Seriola lalandi. J World Aquaculture Soc. 49(1): 113-126.
Bergman, A.M., Trushenski, J.T., and Drawbridge, M. 2018. Addition of emulsifiers to hydrogenated soybean oil-based feeds for California Yellowtail. North American Journal of Aquaculture 80(1) 13-21.
Stuart, K., Johnson, R., Armbruster, L. and Drawbridge, M. 2018. Arachidonic acid in the diet of captive California yellowtail Seriola dorsalis and its effects on egg quality. North American Journal of Aquaculture 80:97–106
Fish Health
A comprehensive and viable fish health program is key to the success of any aquaculture enterprise because it extends into all aspects of hatchery operations at some level. HSWRI maintains a Fish Health Management Team that meets routinely to discuss and evaluate issues related to fish health. HSWRI’s approach to aquaculture begins with disease prevention, which is only possible when the culture requirements of the animal are well understood and accommodated to every extent possible, or when best management practices are employed for new species.
Research in the area of fish health at HSWRI has been varied over the years but can generally be categorized into 1) improving fish survival and quality, 2) identifying pathogens in cultured animals and determining appropriate treatment options, and 3) surveying wild fish populations to be able to understand their health status and relate it to the species in culture. The importance of this work cannot be overstated relative to fisheries replenishment programs where cultured fish are intentionally released into the wild, and the corollary concerns surrounding accidental escapement of farmed fish from ocean net pens.
Recently, we have instituted a semi-quantitative surveillance program of documenting skeletal malformations in larval and juvenile finfish at our marine hatchery as part of our fisheries replenishment program. Goals of this surveillance program are to document cultured fish malformation incidence, assess morphologic variation between wild and cultured finfish species, and prevent the release of a hatchery fish phenotype. Disease prevention and diagnosis is an important component for all finfish life stages, ranging from larvae to broodstock. We consider all aspects of species history, environment, water quality, nutrition, husbandry, or stress as they may interact with potential pathogens that can result in finfish disease. We institute surveillance programs to track early indicators of non-infectious disease agents such as gas bubble disease, which are difficult to treat and can result in long-term vision impairment. We focus on strong biosecurity to prevent pathogen introduction and seek to improve animal welfare using new anesthetics with minimal withdrawal times, and to address the public health and safety of seafood consumers.
Published Research
Silbernagel, C., Shane, M., Drawbridge, M. (in prep) Quality considerations and malformation surveillance in a marine stocking program.
Stevens, B., Gorges, A., Silbernagel, C. 2020. Efficacy of two different doses of 10% eugenol in adult California yellowtail (Seriola dorsalis). Aquaculture Research 51: 1753-1756.
Bowker, J., Wandelear, N., Silbernagel, C. 2017. Use of Aqui-S 20E to sedate California yellowtail and white seabass to handleable. U.S. Fish & Wildlife Service AADAP Drug Research Information Bulletin, DRIB No. 54.
Silbernagel, C. and Yochem, P. 2016. Effectiveness of the anesthetic Aqui-S 20E in marine finfish and elasmobranchs. Journal of Wildlife Diseases 52(1) Supplement: S96-S103.
Curtis, P. A., M. A. Drawbridge, T. Iwamoto, T. Nakai, R. P. Hedrick, and A. P. Gendron. 2001. Nodavirus infection of juvenile white seabass cultured in southern California: first record of viral nervous necrosis (VNN) in North America. Journal of Fish Diseases. 24:263-271.
Stuart, K.R., M. Keller, and M. Drawbridge. 2010. Efficacy of formalin and povidone-iodine disinfection techniques on the eggs of three marine finfish species. Aquaculture Research. Vol. 41(11): 838-843.
Smiley, J.E., M.A. Drawbridge, M.S. Okihiro, and R.S. Kaufmann. 2011. Acute effects of gas supersaturation on juvenile cultured white seabass (Atractoscion nobilis). Trans Am Fish Soc. 140:1269-1276.
Smiley, J.E., M.A. Drawbridge, M.S. Okihiro, and R.S. Kaufmann. 2012. Pathology of ocular lesions associated with gas supersaturation in white seabass (Atractoscion nobilis). Journal of Aquatic Animal Health 24:1-10.
Stuart, K., F. Rotman, and M. Drawbridge. 2015. Methods of Microbial Control in Marine Fish Larval Rearing: Clay-based Turbidity and Passive Larval Transfer. Aquaculture Research DOI: 10.1111/are.12696
Mahboudi, F. G. 1987. In vivo and in vitro immunization of White Seabass (Atractoscion nobilis) with Vibrio parahaemolyticus O-antigen. Master’s Thesis, San Diego State University. 90 pp.
Kavadellas, A. 2006. The parasitic diseases and state of internal organs of farmed, released and recaptured white seabass (Atractoscion nobilis). PhD Thesis, University of Portsmouth
Fish Physiology
Research on fish physiology at HSWRI has ranged widely and often involved outside collaborators. Understanding of physiological processes in cultured organisms is a component of fish health that is critical to optimizing husbandry conditions. Until various physiological processes can be studied in detail, culturists often rely on trying to replicate the natural conditions those life stages are found to the extent that they are known and can be simulated. Examples of physiological research we have been involved in can be gleaned from the reference list below. This includes studies of the effects of gas supersaturation in juvenile white seabass, which are particularly susceptible. Concurrent collaborative studies on the effects of exercise conditioning on growth, muscle development and stress tolerance also fall within this discipline. Not well represented by our publication list are a series of studies designed to define the thermal preferences and tolerance limits of various species and life stages under culture. This information is needed to optimize thermal regimes in recirculating systems where temperature is controlled and to avoid exposing species and life stages to temperature extremes in systems that cannot be controlled such as flow-through tanks or ocean cages.
Published Research
Smiley, J.E., M.A. Drawbridge, M.S. Okihiro, and R.S. Kaufmann. 2011. Acute effects of gas supersaturation on juvenile cultured white seabass (Atractoscion nobilis). Trans Am Fish Soc. 140:1269-1276.
Smiley, J.E., M.A. Drawbridge, M.S. Okihiro, and R.S. Kaufmann. 2012. Pathology of ocular lesions associated with gas supersaturation in white seabass (Atractoscion nobilis). Journal of Aquatic Animal Health 24:1-10.
Wegner, N.C., Drawbridge, M.A. and Hyde, J.A. 2018. Reduced swimming and metabolic fitness of aquaculture-reared California Yellowtail (Seriola dorsalis) in comparison to wild-caught conspecifics. Aquaculture 486 (2018): 51–56
Cepuritas, A. (2005). Exercise effects on growth rate, IGF activity, and cortisol in juvenile White Seabass. Master’s Thesis, University of San Diego.
Peters, C. (2009). Enhancement of growth rates and swimming performance in juvenile marine finfish in aquaculture. Master’s Thesis, University of San Diego.
Buhr, G. (2002). The effects of exercise conditioning on the growth and development of juvenile White Seabass (Atractoscion nobilis). Master’s Thesis, University of San Diego.
Swaney, J. (2002). Effects of temperature and ration levels on growth of White Seabass, Atractoscion nobilis. Master’s Thesis, University of San Diego. 97 pp.
True, C. D. 1994. Influence of different salinities and temperatures on the metabolism and osmoregulation in juvenile White Seabass, Atractoscion nobilis. Master’s Thesis, Autonomous University of Baja California, Ensenada, Mexico (in Spanish). 62 pp.
Schwebel, L.N., K. Stuart, M.S. Lowery, and Wegner, N.C. 2018. Swim bladder inflation failure affects energy allocation, growth, and feed conversion of California Yellowtail (Seriola dorsalis) in aquaculture. Aquaculture, 497, 117-124.
Schwebel, L.N. 2017. Understanding Growth and Development of California Yellowtail (Seriola dorsalis) in Aquaculture Using Physiological Tools. University of San Diego
Genetics
Genetic tools are applied throughout the Sustainable Seafood Program to improve the culture of organism for fisheries replenishment and seafood production. First, genetic parentage analysis plays an important role in understanding the population dynamics of captive breeders (broodstock) and has been applied extensively by HSWRI with California yellowtail and white seabass. This technique allows cultured offspring to be linked to the parental broodstock, thereby informing decisions that maximize reproductive output and performance (e.g. removing a breeding individual that is producing low quality eggs or larvae). In a fisheries replenishment context, the known genetic makeup of the broodstock has the potential to be used as an identifier of offspring that have been released into the wild. A genetic mark for cultured white seabass is currently being developed that would complement the existing marking approach using coded wire tags. Genetic marking provides a lifetime mark (no tag loss), with no adverse handling or tagging effects, and can be detected non-invasively from a small fin clip. As part of this project, archived biological samples (fin clips and otoliths) will also be used to retrospectively estimate the contribution rate of cultured white seabass to total catch and determine if coded wire tag loss has led to under-detection in the catch.
Fisheries replenishment requires rigorous broodstock management to maintain sufficient levels of genetic variability in cultured offspring intended for release into the wild. For this reason, broodstock of both white seabass and California halibut maintained by HSWRI are composed exclusively of wild-origin individuals and are selected according to best available scientific information on the wild population structure (hatchery breeding groups reflect local sub-populations, where applicable). Protocols have also been developed, using the parentage approach described above, for appropriate broodstock rotation and replacement schedules to meet this objective. For seafood production, broodstock management to maintain genetic diversity is less critical as these offspring will not be released into the wild. Instead, the focus is on selecting for traits of interest (e.g. fast growth, tolerance to specific environmental conditions). Depending on the research application, California yellowtail broodstock held at HSWRI consist of mixed individuals (wild- and cultured-origin) or entirely culture-origin individuals (first and second generation). Currently, a research area of interest is developing cold tolerant strains of California yellowtail that would perform well in cage culture off the coast of southern California.
Published Research
Bartley, D. M., & Kent, D. B. (1990). Genetic structure of white seabass populations from the southern California bight region: applications to hatchery enhancement. California Cooperative Oceanic Fisheries Investment Report, 31, 97-105.
Bartley, D. M., Kent, D. B., & Drawbridge, M. A. (1995). Conservation of genetic diversity in a white seabass hatchery enhancement program in southern California. American Fisheries Society Symposium 15:249-258
Craig, M., Purcell, C., Stuart, K., Drawbridge, M., & Hyde, J. Uses of genetic parentage analysis in cultured California yellowtail (Seriola dorsalis) (2017). In Genetics and Breeding in Aquaculture: Proceedings of the 44th U.S.-Japan Aquaculture Panel Symposium, Seattle, WA, November 1, 2016. U.S. Dept. Commerce, NOAA Tech.Memo. NMFS-F/SPO-175, 78 p.
Gruenthal, K. M., & Drawbridge, M. A. (2012). Toward responsible stock enhancement: broadcast spawning dynamics and adaptive genetic management in white seabass aquaculture. Evolutionary Applications, 5(4), 405-417.
Gruenthal, K. M., Gauger, B. J., & Drawbridge, M. A. (2014). Maternal reproductive exhaustion in a broadcast spawning marine finfish cultured for conservation. Aquaculture, 422, 129-135.
Purcell, C., Severin, A., Buonaccorsi, V. P., Drawbridge, M., Stuart, K., & Hyde, J. (2017). RNAseq analysis of early larval development in Seriola lalandi with emphasis on differential development of digestive system in fast and slow growing groups. In Genetics in Aquaculture: Proceedings of the 42nd U.S.-Japan Aquaculture Panel Symposium, La Jolla, CA, October 1, 2014. U.S. Dept. Commerce, NOAA Tech. Memo. NMFS-F/SPO-168, 87 p.
Purcell, C., Severin, A., Drawbridge, M., Stuart, K., & Hyde, J. (2017). The de novo draft assembly of the yellowtail, Seriola dorsalis, genome. In Genetics in Aquaculture: Proceedings of the 42nd U.S.-Japan Aquaculture Panel Symposium, La Jolla, CA, October 1, 2014. U.S. Dept. Commerce, NOAA Tech. Memo. NMFS-F/SPO-168, 87 p.
Purcell, C., Wegner, N., Drawbridge, M., Stuart, K., & Hyde, J. (2017). Genetic and physiological studies of health and fitness in aquaculture-reared California yellowtail, Seriola dorsalis. In Genetics in Aquaculture: Proceedings of the 42nd U.S.-Japan Aquaculture Panel Symposium, La Jolla, CA, October 1, 2014. U.S. Dept. Commerce, NOAA Tech. Memo. NMFS-F/SPO-168, 87 p.
Severin, A., Purcell, C., Drawbridge, M., Masonbrink, R., Seetharam, A., Ozaki, A., Araki, K., Usuki, H., Okuzawa, K., Ototake, M., Ito, F. & Hyde, J. (2017) Seriola genomics and the knowledge repository serioladb. org. In Genetics and Breeding in Aquaculture: Proceedings of the 44th U.S.–Japan Aquaculture Panel Symposium, Seattle, WA, November 1, 2016. U.S. Dept. Commerce, NOAA Tech.Memo. NMFS-F/SPO-175, 78 p.
Smith, E. (2015). Evaluation of reproductive strategies in captive California Yellowtail (Seriola lalandi) using genetic parentage analyses. Master’s Thesis, University of San Diego.
Smith, E., Stuart, K., Hyde, J., Purcell, C., Morse, G., & Drawbridge, M. (In press). Spawning dynamics and egg production characteristics of captive Seriola dorsalis assessed using parentage analyses. Aquaculture.
Advanced technologies and engineering
As our production systems have moved from flow-through to recirculated, and scales of production have increased from experimental to commercial levels, the need to increase the efficiency and sophistication of systems has also increased. As with many facets of aquaculture, life support systems technologies are continuously evolving. Both HSWRI aquaculture facilities serve as testing grounds for new technologies and combinations of system components. Gradually, component preferences are evolving for particular species and life stages. Among the desired, if not required, attributes of these components are simplicity, durability (in seawater), reliability, effectiveness and efficiency. In addition to “standard” recirculation system components, creative design approaches are being applied to develop species-specific systems for collecting brood fish, integrated fish grading and transfer, tagging, live feed production and delivery, monitoring and control, and integrated species systems. We have also tested various cage designs in the offshore environment, including a submersible cage system.
One area of innovative research for us has centered on finding ways to mitigate gas imbalances in the environment and its effects on fish. This has included developing portable hyperbaric chambers to offset barotrauma in brood fish collected from deep water, as well as customizing efficient but relatively inexpensive vacuum degassing systems to offset supersaturated seawater conditions that result in gas bubble disease. Our hyperbaric chambers are now being used and replicated by other researchers and public aquariums to study and collect fish successfully from deep water. Our research on chronic effects of supersaturation in white seabass continues and effective vacuum degassers are in the final stages of development and implementation.
Another recent example of our design and testing capacity is a larval tank project that we conducted in collaboration with Oceans Design, Inc. In this project we developed a state-of-the-art, commercial-scale, self-cleaning larval tank that is now in commercial production. The basic premise of this tank design was to include a motor-driven cleaning arm that continuously “sweeps” debris off the bottom and walls of the tank and into a trench that spans the radius of the tank. This project was very successful and has led to an ability for owners to grow more fish per unit area with less labor.
We are also currently expanding our fish culture to include marine algae and invertebrates in both flow through and seawater reuse configurations. These different organisms are being combined in a rearing system in what is referred to as integrated multi-trophic aquaculture or simply IMTA. In these systems, resource inputs are maximized as downstream trophic levels utilize solid and dissolved nutrients from the upstream organisms. Waste solids can be 35% of the feed given to the fish, so recapturing this in the form of edible invertebrates represents a significant gain. Furthermore, by removing dissolved nitrogen and phosphorus from the system, macroalgae reduce the requirements for whole-system water exchange and thereby reduce effluent volume. This opens up possibilities for siting new farms and the information gained will be useful to growers who may be interested in diversifying their product line of seafood in an efficient manner that maximizes resource inputs.
Published Research
Smiley, J.E. and M.A. Drawbridge. 2007. Techniques for live capture of deepwater fishes with special emphasis on the design and application of a low-cost hyperbaric chamber. Journal of Fish Biology 70:867-878.
Smiley, J.E. and M. Drawbridge. 2008. A simple apparatus for maintaining gas supersaturated seawater in the laboratory for experimental purposes. North American Journal of Aquaculture. 70:61–67.
Stuart, K., Paquette, M., & Drawbridge, M. (2009). Integrated larval-nursery system for California halibut. Global aquaculture advocate.
Development of spawning and larval culture techniques
While this is not an obvious or traditional discipline of biology, within an aquaculture setting it is the basis for success or failure for any given species. Someone in the supply chain needs to be able to reliably produce mass quantities of high quality fingerlings for growout. Achieving this requires successfully spawning adults and rearing the larvae – two components of the culture process that often represent the greatest bottlenecks. The importance of quality cannot be overstated when producing any life stage from egg to fingerling. We have successfully spawned and reared a range of species, including being the first, and in cases the only, to successfully rear fingerlings of some species. We have developed mass production techniques for California yellowtail, California halibut, and white seabass to the extent that these species could be commercialized today. We have also successfully bred and cultured other species like California sheephead, starry rockfish, cabezon, kelp bass, and giant sea bass on an experimental basis. While each species has their nuances when it comes to culture, many techniques that we have developed can be applied to other species to greatly reduce the learning and start-up curve.
Published Research
Jirsa, D., M. Drawbridge, and K. Stuart. 2007. Spawning of a captive population of California sheephead, Semicossyphus pulcher. Journal of the World Aquaculture Society 38(1): 122-128.
Jirsa, D., M. Drawbridge, and K. Stuart. 2009. The effect of tank color and light intensity on growth, survival, and stress tolerance of white seabass, Atractoscion nobilis, larvae. Journal of World Aquaculture Society 40(5): 702-710.
Stuart, K., and M. Drawbridge. 2011. The effect of light intensity and turbidity on survival and growth of cultured larval yellowtail amberjack (Seriola lalandi). Aquaculture 321: 152-156.
Stuart, K., and M. Drawbridge. 2012. The effect of photoperiod on larval culture performance of two marine finfish species. Aquaculture 360-361: 54-57.
Stuart, K., and M. Drawbridge. 2013. Captive spawning and larval rearing of California yellowtail (Seriola lalandi). Aquaculture Research 44(5): 728-737.
Stuart, K.R. 2013. Chapter 2: The effect of light on larval rearing of marine finfish. Pp. 25-40 in J.G. Qin (ed.) Larval Fish Aquaculture. Nova Science Publishers, Hauppauge, NY.
Stuart, K.R., L. Armbruster, R. Johnson, and M.A. Drawbridge. 2020. Egg diameter as a predictor for egg quality of California yellowtail (Seriola dorsalis). Aquaculture 522, 735154.
Environmental monitoring
The environmental monitoring program at HSWRI has evolved from dual needs. First, in initiating scientific studies of recirculation systems and associated components, it became clear that more sophisticated testing methods, yielding greater resolution and precision than typical colorimetric and photometric methods, would be desired. Additionally, it was desirable to develop methodologies for accurately measuring less common or more technically challenging parameters in seawater (e.g. Total Volatile Solids, silicates, phosphates). Even if samples are not tested in-house, coordination of sample collection and testing by outside laboratories is often required.
The second area of need has been for environmental monitoring of seawater and sediment quality associated with net pens. Toward this end, well established monitoring protocols for salmon farms in the northwest were adapted to Southern California through training and mentoring of HSWRI staff by experts from that area. Although not mandated by any regulations due to the small scale of HSWRI operations, having the techniques well established and having obtained baseline data from the net pens is proving to be extremely valuable. Coupled with this capability to monitor existing farm sites is research directed toward selecting appropriate new sites, which involves working collaboratively with modelers and collecting pertinent data such as sediment characteristics and seasonal current information using acoustic Doppler current meters.
Published Research
Brooks, K.M., 2007. Sediment physicochemical monitoring at delayed release netpens and raceways for white seabass located in Southern California during the period 2004 through 2006. Report prepared for Hubbs-SeaWorld Research Institute (HSWRI), California Department of Fish and Game (CDFG), and Advisors to the Ocean Resources Enhancement and Hatchery Program (OREHP), 119pp.
Drawbridge, M. A., P. Sylvia, and M. A. Shane. 2009. Offshore culture of California Yellowtail (Seriola lalandi) and Striped Bass (Morone Saxatilis): A multinational demonstration project supported by a well-established tuna farming infrastructure. Final Report for Project #NA06OAR4170249 (Sept 2006- November 2009), 81pp.
Fish tagging and tracking
The marking or tagging of fish is a fundamental fishery tool that our scientists employ to identify or differentiate our cultured fish from other individuals or groups of fish. Since the late 1980’s the research we have conducted under our fisheries replenishment program has utilized various marking methods (e.g. streamer or spaghetti tags, chemical dyes, elastomer compounds) for our cultured fish that are released into the wild. Additionally, these tagging methods are also used in tank studies to identify individuals as well as understand tag retention. The most common tagging method that we employ in our replenishment program for white seabass, is the insertion of a tiny coded wire tag into the cheek musculature of each fish. Each 1.1 mm length tag has a unique code etched on it that helps our scientists identify the hatchery-reared fish upon its recapture. This tagging technology has helped the program to identify patterns of movement, dispersal, survival as well as wild prey items of released cultured white seabass through our fisheries independent and dependent sampling efforts. We have recaptured white seabass that have been at liberty, in the Pacific Ocean, from just several days up to 20 years. We have also recaptured them 160 km (100 miles) off the coast of San Diego and more than 550 km (340 miles) near the coast from their release location.
Acoustic telemetry is another method that HSWRI researchers employ to understand the fine scale movements and dispersal of fish. We implement this technology by surgically implanting a small acoustic transmitter (sound producing device) into the abdomen of fish. Each transmitter generates a slightly different sound pattern that permits investigators to identify individual fish. When fish are released into the wild their movements can be tracked either actively or passively. When actively tracking, an underwater hydrophone (listening device) is deployed from a small vessel so that investigators can hear and follow the fish. Passive monitoring utilizes moored underwater receivers that can detect, identify and store data on the receiver when a fish with a transmitter swims near them. These receivers are deployed in various arrays in embayments and along the coastline. Periodically HSWRI scientists retrieve and redeploy the receivers to download the data. Some of the interesting science we have learned from using this technology includes identifying predators and patterns of emigration from embayments for the cultured white seabass we are releasing into the ocean.
Published Research
Allen, L. G., Pondella II, D. J., & Shane, M. A. 2007. Fisheries independent assessment of a returning fishery: Abundance of juvenile white seabass (Atractoscion nobilis) in the shallow nearshore waters of the Southern California Bight, 1995–2005. Fisheries Research, 88(1-3), 24-32.
Drawbridge, M. A., Kent, D. B., Shane, M. A., & Ford, R. F. 1995. The assessment of marine stock enhancement in southern California: A case study involving the white seabass. American Fisheries Society, 15: 568-569.
Hervas, S., Lorenzen, K., Shane, M. A., & Drawbridge, M. A. 2010. Quantitative assessment of a white seabass (Atractoscion nobilis) stock enhancement program in California: Post-release dispersal, growth and survival. Fisheries Research, 105(3), 237-243.
Population biology and modeling
A thorough understanding of the target species biology, both wild and cultured, is crucial for fisheries replenishment efforts to be successful. Sampling programs provide key information (e.g. growth, fecundity, natural mortality) needed to effectively design hatchery and release operations. An extension of this is population modeling, which is becoming an increasingly useful tool in fisheries replenishment science. Models, informed using real-world data (e.g. from sampling programs or laboratory studies), are used for a variety of applications including forecasting the potential outcomes of releasing cultured fish into the wild (e.g. feasibility planning), and for evaluating and adaptively managing ongoing replenishment efforts. For example, several years of mark-recapture information from scientific gillnet surveys were used to model post-release survival in cultured white seabass, allowing optimal release strategies to be identified and implemented for this species. HSWRI works closely with researchers at the University of Florida to apply population modeling in its fisheries replenishment programs for white seabass and California halibut.
Published Research
Donohoe CJ. 1997. Age, growth, distribution, and food habits of recently settled White Seabass, Atractoscion nobilis, off San Diego County, California. Fish Bull 95(4):709-21.
Allen, L. G., Pondella II, D. J., & Shane, M. A. (2007). Fisheries independent assessment of a returning fishery: Abundance of juvenile white seabass (Atractoscion nobilis) in the shallow nearshore waters of the Southern California Bight, 1995–2005. Fisheries Research, 88(1-3), 24-32.
Drawbridge, M. A., Kent, D. B., Shane, M. A., & Ford, R. F. 1995. The assessment of marine stock enhancement in southern California: A case study involving the White Seabass. American Fisheries Society, 15: 568-569.
Hervas, S., Lorenzen, K., Shane, M. A., & Drawbridge, M. A. (2010). Quantitative assessment of a white seabass (Atractoscion nobilis) stock enhancement program in California: Post-release dispersal, growth and survival. Fisheries Research, 105(3), 237-243.
MacNamara, R., Camp, E., Shane, M., Lorenzen, K., & Drawbridge, M. (In prep) Modeling release strategies and bioeconomic tradeoffs in fisheries stock enhancement of a coastal flatfish Paralichthys californicus.
Biological and ecological applications using cultured fish
Aquaculture of plants and animals is most commonly associated with the production of food, similar to agriculture on land. But of course, there are other applications and market outlets such as providing aquatic organisms for the pet trade, for scientific and medical applications, for bait, etc. Depending on the organism in question, aquaculture can offer access to study subjects that would otherwise be very difficult, if not impossible, to study in the wild. For example, eggs and larvae of marine fishes are challenging to source from the wild and perhaps even more so to maintain alive. In an aquaculture setting, these life stages can be made available with regularity and often in great numbers depending on the species. The fact that cultured organisms can be used to help benefit management of the wild stocks and the overall health of our oceans, creates an excellent opportunity that should be fully exploited. Examples of contemporary applications include investigations of the effect of climate change; ocean acidification; contaminants, including oil spills; barotrauma associated with catch and release; and sound pollution.
Published Research
Aalbers, SA, Stutzer GM, Drawbridge MA. 2004. The effects of catch-and-release angling on the growth and survival of juvenile White Seabass captured on offset circle and J- type hooks. N Am J Fish Manage 24(3):793-800.
Aalbers SA. 2008. Seasonal, diel, and lunar spawning periodicities and associated sound production of White Seabass (Atractoscion nobilis). Fish Bull 106(2):143-51.
Gleason LU and Burton RS. 2012. High-throughput molecular identification of fish eggs using multiplex suspension bead arrays. Molecular Ecology Resources 12(1):57-66.
Aalbers SA and Sepulveda CA. 2012. The utility of a long-term acoustic recording system for detecting White Seabass Atractoscion nobilis spawning sounds. J Fish Biol 81(6):1859-70.
Stransky, B.C. 1998. Assessment of sediment quality effects in Mission Bay and San Diego Bay on the growth, behavior and survival of juvenile White Seabass (Atractoscion nobilis) and juvenile California Halibut (Paralichthys californicus). Master’s Thesis, San Diego State University. 287 pp.
Shen SG, Chen F, Schoppik DE, Checkley DM Jr (2016) Otolith size and the vestibulo-ocular reflex of larvae of white seabass Atractoscion nobilis at high pCO2. Mar Ecol Prog Ser 553:173-183. https://doi.org/10.3354/meps11791
Coffin, S., Magnuson, J.T., Volz, D.C., and D. Schlenk. 2020. Effects of short-term exposure to environmentally-relevant concentrations of benzo(a)pyrene-sorbed polystyrene to White seabass (Atractoscion nobilis). Environmental Pollution. 263: 1-7. https://doi.org/10.1016/j.envpol.2020.114617
Conti, Stephane G., Benjamin D. Maurer, Mark A. Drawbridge, and David A. Demer. 2006. Measurements of total scattering spectra from boccacio (Sebastes paucispinis). Fishery Bulletin 105(1), 153-157.
Fish Behavior
Behavioral traits in cultured fish often do not match those of wild fish. This is largely a consequence of conditions in the hatchery (e.g. lack of predators, plentiful food, bare tanks). Though this allows large numbers of healthy fish to be produced efficiently, the opportunity to develop skills necessary for life in the wild is generally lacking. HSWRI scientists seek to understand the behavioral ecology of cultured fish as part of the fisheries replenishment programs; specifically, the extent of behavioral deficits between cultured and wild fish, and what approaches may allow for the development of more “wild-like” behavioral skills in cultured fish intended for release into the wild. HSWRI continues to explore topics such as environmental enrichment, live-prey exposure and release site acclimation to develop behaviorally robust cultured fish and ultimately improve post-release survival in the wild.
Published Research
Dutton P. (1992). Effects of experience on feeding success by larval White Seabass, Atractoscion nobilis. J Fish Biol 41(5):765-73.
Louie, L. (2005). Behavioral comparisons between wild and cultured juvenile California Halibut (Paralichthys californicus). Master’s Thesis, University of San Diego.
Rudolph, J.D. (1995). Feeding and predator avoidance strategies of cultured White Seabass, Atractoscion nobilis. Master’s Thesis, San Diego State University. 106 pp.
Tardy, K. (2011). Functional morphology and swimming behavior in larval and juvenile White Seabass (Atractoscion nobilis). Master’s Thesis, University of San Diego.